Abstract
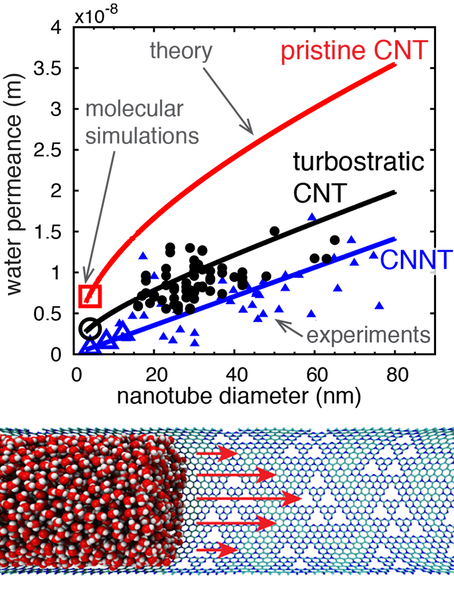
The independent effect of nanotube surface chemistry and structure on the flow of water under nanoscale confinement is demonstrated in this paper for the first time via the synthesis of novel carbon nitride nanotube (CNNT) membranes. Using a combination of experiments and high-fidelity molecular dynamics (MD) simulations, it is shown here that the hydrophilization of the sp2 carbon structure, induced by the presence of the C–N bonds, decreases the pure water permeance in CNNTs when compared with pristine and turbostratic carbon nanotubes (CNTs). The MD simulations are based on a model true to the chemical structure of the synthesized nanotubes, built from spectroscopy measurements and calibrated potentials using droplet experiments. The effect on permeance is explained in terms of solid–liquid interactions at the nanotube wall with increased water viscosity and decreased surface diffusion near the CNNT wall, when compared to CNTs. A model directly linking the solid–liquid interactions to the water permeance is presented, showing good agreement with both experiments and MD simulations. This work opens the way to tailoring surface chemistry and structure inside nanotube membranes for a wide range of transport and separation processes.